Wikipedia:Reference desk/Archives/Science/2013 January 9
| Science desk | ||
|---|---|---|
| < January 8 | << Dec | January | Feb >> | January 10 > |
| Welcome to the Wikipedia Science Reference Desk Archives |
|---|
| The page you are currently viewing is an archive page. While you can leave answers for any questions shown below, please ask new questions on one of the current reference desk pages. |
January 9
[edit]a map showing the international date line in the middle
[edit]I would like to be able to see a world map with the international date-line more centred. (in one piece) - ie: i do not want to see part of the one side of the date-line SQUEEZED in on the right- and the other part SQUEEZED in on the left of the whole image/picture/diagram.
Looking at an existing time-zone map of the world, a more convenient split would be somewhere between 30 to 60 (degrees) West longitudes - as this looks like a longitudinal zone with the least breaks or "zig-zags".
Would you be able to help me, please? 196.210.195.46 (talk) 05:19, 9 January 2013 (UTC)
- Does File:Standard time zones of the world (2012) - Pacific Centered.svg work for you? If not, you can type "Pacific-centered map" into Google Images and get hundreds of options. --Jayron32 05:48, 9 January 2013 (UTC)
Hand grenade
[edit]How many times can a hand grenade realistically bounce off a wall before either coming to a stop or exploding, whichever comes first? Assume that: the grenade in question is a German stick grenade with a standard 5-second fuse, thrown at the wall at a 45 to 60 degree angle as hard as possible; the thrower is tall and has long arms but only average strength; and the walls are concrete and about 3 feet apart, forming a narrow passageway. 24.23.196.85 (talk) 06:46, 9 January 2013 (UTC)
- It could only bounce of a wall once. If you mean bounce between two walls, that would depend on how high along the walls it hit initially, it's angle relative to the ground, and it's initial speed. They don't bounce very much, so it would have to be going quite fast to bounce more than once or twice. StuRat (talk) 06:53, 9 January 2013 (UTC)
- That's what I meant, bouncing between the walls of a concrete-walled passageway. 24.23.196.85 (talk) 07:06, 9 January 2013 (UTC)
- It still depends on the width of the passageway (imagine a passageway 100 feet wide - there could only possibly be one bounce...now imagine a passageway only a foot wide - and there would obviously be many bounces)...also the height at which the grenade initially hits the first wall...if it hit the first wall very low to the ground then fewer bounces than high up. Then we need to know the speed of impact, the angle of the impact...there are just far too many variables.
- Worse still - they have a very irregular shape - how it hit (stick first, head first, sideways) would make a massive difference. The rate and direction of rotation would add a complicating factor. Also the head of the grenade was very thin metal - it would likely dent easily - absorbing energy from that first bounce and drastically reducing it's speed. But if the stick hit first - it's wood, flexible like a very stiff spring - so it would probably retain most of the energy providing more rebound.
- If you want to get even more technical - our article says that the 1942 and later versions had a serrated steel sleeve that could be optionally be slid over the head to get a greater fragmentation effect in anti-personnel applications - which would really complicate matters in a head-first impact because the serrations would result in a much more random bounce/spin.
- Gut feel says "once"...or maybe "not at all" - but there are too many variables to come up with any kind of informed answer.
- If I were you - I'd take a foot of broom-handle and firmly duct-tape a 500g can of soup to the top - then get out there and try it! Nothing short of tossing one around yourself will give you the authentic "feel" you need for how they would bounce.
- (We need pictures! :-)
- SteveBaker (talk) 14:04, 9 January 2013 (UTC)
- Not soup. Anything liquid will slosh and act as a damper. Something solid enough to not slosh when you shake it is better. A can of chili or dog food should do the trick.
- I am guessing that straight on to the wall will give you maybe a one foot bounceback, but a glancing hit will retain most of the velocity -- but make the next wall effectively farther away, so maybe two bounces. I really want to see pics of this. Do it! Do it for SCIENCE!! <smile> --Guy Macon (talk) 15:56, 9 January 2013 (UTC)
- Good point about the liquid thing...but most regular cans are only about 250 to 350 grams - I was thinking specifically of the larger diameter cans that might weigh more like the 560 grams that the real grenade weighed...minus the weight of the stick...so a 500g soup can would have about the right weight. I've seen dogfood in those larger cans though - that would work. SteveBaker (talk) 17:31, 9 January 2013 (UTC)
- @24.23.196.85, are you asking this because you want to incorporate it into a graphic novel, film script, story, or other work of fiction? If so, one, maybe two bounces would be the limit of "realistic". But if you're going for exaggerated violence a la Tarantino, have it bounce as many times as you want! - LuckyLouie (talk) 16:29, 9 January 2013 (UTC)
- (In a followup to a previous question (Wikipedia:Reference_desk/Science#Lever_frame), our OP indicated that, he/she is indeed writing a novel set in WWII).
- I suspect that one bounce (or possibly no significant rebound at all) is the most likely "realistic" outcome - but two bounces and some rolling would seem "credible" to me - especially in a book where your imagination gets to roam free. It might be harder to make it believable in a movie though. Our article on the later Model 43 grenade suggests a detonation delay of 5.5 to 7 seconds...which might help to extend any exciting rolling and/or bouncing and/or being-"fetched"-by-a-stray-dog deemed necessary for the plot. SteveBaker (talk) 17:28, 9 January 2013 (UTC)
- As long as we're working in fiction, maybe have the grenade thrower execute a complicated bank shot off the wall in order to take out the bad guy. - LuckyLouie (talk) 18:36, 9 January 2013 (UTC)
- That was my plan all along -- a bank shot to send the potato-masher behind a partition in order to take out a German machine-gunner. Based on the layout of that particular pillbox, I can do it with two bounces, but I'll have to shorten the partition somewhat to make it happen. :-) BTW, I like the experiment you suggested, but the one thing to which I don't have access is concrete walls 3 feet apart -- all the walls around me are made of wood. Will that make a significant difference? 24.23.196.85 (talk) 00:19, 10 January 2013 (UTC)
- I find it really hard to believe that you'd have the time or information to plan that kind of a trick. Pillboxes had all sorts of internal layouts - and it's hard enough to toss the grenade into the firing slit as it is without having to consider bounce angles and all of that stuff. The stick grenade is particularly ill-suited to that kind of trickery anyway because it's such a weird shape and really off-balance! It's not like you can stand a couple of feet outside the firing slit, examine where the internal walls are, consider the angles, wind up for a really accurate toss with just the right spin, speed and direction! There are a bunch of guys inside who are working very hard to be sure that you don't toss ANYTHING in there! You'd be standing with your back to the concrete wall, with grenade in hand - and consider yourself lucky if you could get the thing inside in any way at all without having your hand blown off! It's also very dark inside those pillboxes - it would be tough to discern anything much about the interior structure from outside. So for me, your story's "credibility" is blown long before we have to consider whether the grenade could physically bounce as intended! SteveBaker (talk) 15:08, 10 January 2013 (UTC)
- I love it. Even assuming a Jason Bourne-like hero, it seems there's a lot more practical issues to consider in this scenario than just 'could a stick grenade bounce twice'. I think the OP might be envisioning a door at the back of the pillbox with a partition separating the entryway from the front gun port area. Even so I'm sure it has a metal door the German crew keeps closed. But let's say the door is open or blown off. Much easier to shoot the guard (if there is one) and then run inside blasting your Sten gun or whatever, using the partition for cover. Any way you slice it, this is all fantasy, so why restrict fiction to 'what can be tested'? - LuckyLouie (talk) 17:40, 10 January 2013 (UTC)
- Sure, there are times when the needs of good fiction override practical reality. However, when something really ridiculous happens, it can also detract from the story. (I could cite countless movies and TV shows that were ruined for me by some ridiculous - and unnecessary - impossible thing.) So if there is no loss of narrative quality, one should err on the side of real-world possibility. Clearly our OP's careful questioning of such minutia as the color of the handles in the signal box that our hero uses to redirect the train - suggest that this will be a very grounded-in-reality kind of a novel. In that case, surely there is another clever & creative way to take out this pillbox without our hero doing such a truly super-human trick. Perhaps start with a document like this one: http://www.lonesentry.com/articles/pillboxwarfare/index.html - which describes how US soldiers would approach a pillbox. It provides the valuable advice that a white-phosphorus grenade is more effective against a pillbox than a fragmentation grenade. But it also adds crucial advice about NOT attacking the rear door and that intersecting fields of fire from adjacent pillboxes is a major threat. There is a lot of scope for our hero to be heroic beyond having the ability to bounce a weird shaped grenade off of half-seen walls through a small slit. SteveBaker (talk) 18:12, 10 January 2013 (UTC)
- Not a small slit -- a bent entrance-type passageway that serves as the entry point for the pillbox, which is covered by an MG-42 emplaced in a gunport in the partition around which the passage doubles-back (which machine-gun has to be taken out because it also covers main route of advance). In effect, the passageway leads up a short staircase toward the wall with the machine-gun, then makes a 90-degree left turn for a slightly shorter distance, and doubles back into the pillbox itself. And they don't have to worry about interlocking fire because it's a stand-alone pillbox covering the entrance to a railroad cutting. (If you like to play the Medal of Honor: Allied Assault computer game, at one point you encounter a similar situation in the Normandy level.) 24.23.196.85 (talk) 02:29, 11 January 2013 (UTC)
- Just read the document -- what it says is, rather than not attacking the rear door at all, that the attacker must not stand directly in front of it, because there's always a machine gun covering that sector. But that's not my plan in the first place -- I'm not so stupid as to have my hero stand right in front of the doorway in full view of the machine-gunner. That's why I asked about bouncing the grenade off the walls, because the assault team will have to throw it in while standing to one side of the doorway (which means they're throwing essentially "blind") and try to have it it bounce around the corner to land behind the partition. 24.23.196.85 (talk) 02:44, 11 January 2013 (UTC)
- Sure, there are times when the needs of good fiction override practical reality. However, when something really ridiculous happens, it can also detract from the story. (I could cite countless movies and TV shows that were ruined for me by some ridiculous - and unnecessary - impossible thing.) So if there is no loss of narrative quality, one should err on the side of real-world possibility. Clearly our OP's careful questioning of such minutia as the color of the handles in the signal box that our hero uses to redirect the train - suggest that this will be a very grounded-in-reality kind of a novel. In that case, surely there is another clever & creative way to take out this pillbox without our hero doing such a truly super-human trick. Perhaps start with a document like this one: http://www.lonesentry.com/articles/pillboxwarfare/index.html - which describes how US soldiers would approach a pillbox. It provides the valuable advice that a white-phosphorus grenade is more effective against a pillbox than a fragmentation grenade. But it also adds crucial advice about NOT attacking the rear door and that intersecting fields of fire from adjacent pillboxes is a major threat. There is a lot of scope for our hero to be heroic beyond having the ability to bounce a weird shaped grenade off of half-seen walls through a small slit. SteveBaker (talk) 18:12, 10 January 2013 (UTC)
- I love it. Even assuming a Jason Bourne-like hero, it seems there's a lot more practical issues to consider in this scenario than just 'could a stick grenade bounce twice'. I think the OP might be envisioning a door at the back of the pillbox with a partition separating the entryway from the front gun port area. Even so I'm sure it has a metal door the German crew keeps closed. But let's say the door is open or blown off. Much easier to shoot the guard (if there is one) and then run inside blasting your Sten gun or whatever, using the partition for cover. Any way you slice it, this is all fantasy, so why restrict fiction to 'what can be tested'? - LuckyLouie (talk) 17:40, 10 January 2013 (UTC)
- I find it really hard to believe that you'd have the time or information to plan that kind of a trick. Pillboxes had all sorts of internal layouts - and it's hard enough to toss the grenade into the firing slit as it is without having to consider bounce angles and all of that stuff. The stick grenade is particularly ill-suited to that kind of trickery anyway because it's such a weird shape and really off-balance! It's not like you can stand a couple of feet outside the firing slit, examine where the internal walls are, consider the angles, wind up for a really accurate toss with just the right spin, speed and direction! There are a bunch of guys inside who are working very hard to be sure that you don't toss ANYTHING in there! You'd be standing with your back to the concrete wall, with grenade in hand - and consider yourself lucky if you could get the thing inside in any way at all without having your hand blown off! It's also very dark inside those pillboxes - it would be tough to discern anything much about the interior structure from outside. So for me, your story's "credibility" is blown long before we have to consider whether the grenade could physically bounce as intended! SteveBaker (talk) 15:08, 10 January 2013 (UTC)
- That was my plan all along -- a bank shot to send the potato-masher behind a partition in order to take out a German machine-gunner. Based on the layout of that particular pillbox, I can do it with two bounces, but I'll have to shorten the partition somewhat to make it happen. :-) BTW, I like the experiment you suggested, but the one thing to which I don't have access is concrete walls 3 feet apart -- all the walls around me are made of wood. Will that make a significant difference? 24.23.196.85 (talk) 00:19, 10 January 2013 (UTC)
- As long as we're working in fiction, maybe have the grenade thrower execute a complicated bank shot off the wall in order to take out the bad guy. - LuckyLouie (talk) 18:36, 9 January 2013 (UTC)
- I am guessing that straight on to the wall will give you maybe a one foot bounceback, but a glancing hit will retain most of the velocity -- but make the next wall effectively farther away, so maybe two bounces. I really want to see pics of this. Do it! Do it for SCIENCE!! <smile> --Guy Macon (talk) 15:56, 9 January 2013 (UTC)
- Does it really matter that it's a stick grenade? The German army also issued "egg" grenades (Model 39 grenade) - and it seems much more credible that one of those would bounce and roll around much more than the ungainly stick models. The article on the Model 24 grenade actually says that one of the benefits of the stick design was to prevent it from rolling on hilly terrain - so if the plot requires that the grenade take some complicated path to it's target - then an egg grenade would be a better choice. SteveBaker (talk) 17:44, 9 January 2013 (UTC)
- As a matter of fact, it does matter -- I've never heard of a Maquisard using an egg grenade (unless you meant a pineapple grenade or a Mills bomb). 24.23.196.85 (talk) 06:59, 10 January 2013 (UTC)
- I presume they'd be "liberating" those weapons from the Germans along the way...how else would they have gotten hold of the stick-grenade variety? If they stole the weapons somehow - then they'd be likely to find an occasional "egg" grenade along with the "stick" variety. But if they did that, they'd have to be careful - one sneaky trick the Germans employed when retreating from a position was to swap out the 7 second fuses for 1 second fuses and leave the grenades lying around for the enemy to pick up and attempt to re-use! Yikes! (Of course the hero in a novel would likely know all about this trick and be prepared to re-fuze the grenades before attempting to use them!) SteveBaker (talk) 15:08, 10 January 2013 (UTC)
- Hey, that's an interesting idea -- maybe Blanche's jealous ex-boyfriend will acquire some instant-acting Model 39s from a crooked arms dealer and give them to Mike, hoping he'd blow himself up? ;-) 24.23.196.85 (talk) 02:49, 11 January 2013 (UTC)
- Hmmm - our article Maquis des Glières and Maquis du Haut-du-Bois both say that the British air-dropped large numbers of "mills-bomb" grenades for the use of the Maquis. (One drop is mentioned as containing 150 grenades). So they most certainly did have 'egg'- and 'pineapple'-style grenades. It's entirely possible that they used the stick variety if they could steal them from the Germans...but the British hadn't made stick grenades since 1908 so whatever the Maquis got in air-drops in 1944 would have been "pineapple" fragmentation grenades for sure. It seems unlikely that the Brits would have air-dropped grenades if the Maquis had a good supply of German grenades - so it seems highly likely that pineapple grenades would have been in the majority. Whether they'd prefer stick grenades over pineapples is debatable - stick grenades can be thrown further - but they are a pain to transport and difficult to use in confined spaces. For taking out a pillbox with a fancy rebounding throw, a pineapple grenade would be greatly preferred. SteveBaker (talk) 15:36, 11 January 2013 (UTC)
- Hey, that's an interesting idea -- maybe Blanche's jealous ex-boyfriend will acquire some instant-acting Model 39s from a crooked arms dealer and give them to Mike, hoping he'd blow himself up? ;-) 24.23.196.85 (talk) 02:49, 11 January 2013 (UTC)
- I presume they'd be "liberating" those weapons from the Germans along the way...how else would they have gotten hold of the stick-grenade variety? If they stole the weapons somehow - then they'd be likely to find an occasional "egg" grenade along with the "stick" variety. But if they did that, they'd have to be careful - one sneaky trick the Germans employed when retreating from a position was to swap out the 7 second fuses for 1 second fuses and leave the grenades lying around for the enemy to pick up and attempt to re-use! Yikes! (Of course the hero in a novel would likely know all about this trick and be prepared to re-fuze the grenades before attempting to use them!) SteveBaker (talk) 15:08, 10 January 2013 (UTC)
- As a matter of fact, it does matter -- I've never heard of a Maquisard using an egg grenade (unless you meant a pineapple grenade or a Mills bomb). 24.23.196.85 (talk) 06:59, 10 January 2013 (UTC)
North Pole & South Pole questions
[edit]Is it true that sunrise and sunset only happened once a year in the poles? Why these 2 places got so cold? roscoe_x (talk) 08:10, 9 January 2013 (UTC)
- 1) Kind of. If the horizon is absolutely flat, then yes. However, hills or mountains or even snow drifts mean that as the Sun corkscrews up and down it will pass behind them and then come back out, so you will get multiple "sunsets" and "sunrises".
- 2) It's so cold in summer because the Sun is at such a shallow angle. In winter, it's far colder because there's no sunlight at all, for months at a time. Also, prevailing wind direction is East-West, meaning very little warm air moves in from warmer areas. StuRat (talk) 08:14, 9 January 2013 (UTC)
- Where is East (or West) when standing on any of the poles? bamse (talk) 19:28, 9 January 2013 (UTC)
- At the south pole east is clockwise when looking down and west is anticlockwise. Opposite at the north pole. So in winter time you will not see stars rise in the east and set in the west, but they will just circle you. The point is that the wind does not go towards you to carry heat from outside. Graeme Bartlett (talk) 20:24, 9 January 2013 (UTC)
- Where is East (or West) when standing on any of the poles? bamse (talk) 19:28, 9 January 2013 (UTC)
- That's the point, the winds blow in a circle around the poles (more or less), not over them, where they would deliver warmer air. StuRat (talk) 20:21, 9 January 2013 (UTC)
- So in summer, the sun is at what angle? How low is it? So if the sun will not set, we can see the sun 24 hours a day and we will see it circling in the sky? roscoe_x (talk) 00:43, 10 January 2013 (UTC)
- Yes, it circles for around 6 months. StuRat (talk) 00:45, 10 January 2013 (UTC)
- When the sun is directly above the equator, it appears from the North Pole at the horizon. At the height of summer for the Northern Hemisphere the sun is about 23° north of the equator, so when standing on the North Pole the sun will be seen circling between 0 and 23° above the horizon (i.e. in the summer season). - Lindert (talk) 09:36, 10 January 2013 (UTC)
- Just to clarify that last sentence of Lindert, on any one day the sun will circle at a particular number of degrees above the horizon. That number of degrees will be about 23 (closer to 23 1/2 actually) at the summer solstice, and it will be zero degrees at the autumnal equinox and at the spring equinox. As the days go by after the spring equinox and as we get closer to the summer solstice, the number of degrees above the horizon increases from one day to the next.
- When the sun is directly above the equator, it appears from the North Pole at the horizon. At the height of summer for the Northern Hemisphere the sun is about 23° north of the equator, so when standing on the North Pole the sun will be seen circling between 0 and 23° above the horizon (i.e. in the summer season). - Lindert (talk) 09:36, 10 January 2013 (UTC)
- Here's how to visualize things like this. Get a globe, which will be tilted at 23 1/2 degrees. Put it on a table top so that the interior center of the globe is a certain height above the floor. Hold a thin flashlight (a penlight)(representing the sun) at that same height but some distance away and shine it at the north pole. Spin the globe around to see what is happening during the course of one day. To show the summer solstice, shine the penlight toward the globe from the horizontal direction that the top of the globe is pointing toward. To show one of the equinoxes, shine the light from the same height but after you and the penlight have moved 90 degrees around the globe. You can also look at in-between dates using in-between locations of the penlight. Duoduoduo (talk) 20:16, 10 January 2013 (UTC)
- So the phenomenon is called midnight sun. Okay, so the people in north pole still know the time of day (even at night) by looking at the sun's position relative to objects near the observer? And if I built a house in north pole I wouldn't make a window that will pass sunshine at night time. And another question can you see Aurora from the poles? And if North Pole and North Magnetic Pole is different, also its moving, how far North Magnetic Pole can moved from North Pole? If we make an experiment of flowing water in a tank, will it also moving clockwise in the North Pole? Thanks guys, interesting phenomenon and answers. roscoe_x (talk) 01:41, 11 January 2013 (UTC)
- Here's how to visualize things like this. Get a globe, which will be tilted at 23 1/2 degrees. Put it on a table top so that the interior center of the globe is a certain height above the floor. Hold a thin flashlight (a penlight)(representing the sun) at that same height but some distance away and shine it at the north pole. Spin the globe around to see what is happening during the course of one day. To show the summer solstice, shine the penlight toward the globe from the horizontal direction that the top of the globe is pointing toward. To show one of the equinoxes, shine the light from the same height but after you and the penlight have moved 90 degrees around the globe. You can also look at in-between dates using in-between locations of the penlight. Duoduoduo (talk) 20:16, 10 January 2013 (UTC)
- (outdent) You couldn't build a house at the geographic north pole because it's an ever-shifting (and these days quite unstable) ice floe. So telling the time by the position of the sun or stars would actually be quite difficult. I guess you could use the direction of the magnetic north pole (see below) to provide a fixed line of reference, and from that calculate the time of day in any given time zone. In practice, permanent antarctic stations maintain the time zones of their control centres in nearby southern hemisphere countries. As for the windows: obviously during the nearly-six-month polar night, no window is going to admit light anyway. But when the sun is above the horizon anywhere, Rayleigh scattering makes the whole sky pretty bright. So you'd definitely want blackout curtains, or a bedroom with no windows and only artificial light. (I spent two weeks in the Swedish sub-arctic just after midwinter once; the weirdness of the daylight hours got to me pretty quickly.) I believe the aurora can be seen at the pole. The magnetic poles move pretty slowly, but fast enough that (for example) British Ordnance Survey maps include a magnetic north line, with details on when it was computed, and how far it's expected to deviate in subsequent years. However, in the very long term, it's believed by geologists that the north and south magnetic poles change ends, so the current north magnetic pole could end up arbitrarily close to the south geographic pole. And lastly, unless your experiment is very carefully controlled, the shape of the tank will have more effect than the rotation of the earth. (The earth's rotation does not, for example, affect which was water goes down an ordinary domestic plughole.) Our article Coriolis effect has the details - but the south geographic pole would be a slightly easier place to that experiment, and see the real effect of the earth's rotation on the water, than anywhere else on earth. AlexTiefling (talk) 02:07, 11 January 2013 (UTC)
Why do electrons, nucleons, stars, and planets have spherical shape ?
[edit]I have no microscope to observe electrons, protons, and neutrons in an atom, but I satisfy myself by only looking their pictures in books, on Wikipedia, etc. I usually see that they are spherical. Are they really spherical ? If yes, then, why not other shape ? Why do stars and planets have spherical shape, not other shape ? On the other hand, meteorites are not spherical but irregular. Why ? Parimal Kumar Singh (talk) 09:08, 9 January 2013 (UTC)
- For the astronomical objects, gravity is the culprit. Large enough objects have enough gravity that it crushes anything that sticks up. However, if they rotate quickly, the shape is more of an oblate spheroid. For the subatomic particles, I don't think they really have a shape, just a probability density. It's just simpler to represent them as spheres. See probability amplitude for an alternative representation of an electron in a particular atomic orbital. StuRat (talk) 09:20, 9 January 2013 (UTC)
- That isn't the electron. That's the region where the electron might be found. The electron itself, as far as anyone knows, is a point charge/point mass. Maybe something like string theory would make it something other than a point, but no one knows how to test it. --Trovatore (talk) 09:31, 9 January 2013 (UTC)
- My point is that it's probability envelope is probably the best way to represent it, as that at least has some geometric definition. See atomic orbitals for some others. StuRat (talk) 09:37, 9 January 2013 (UTC)
- I think that's a mistake. Admittedly it's one you see in writing from time to time, but still a mistake. To see the difference, note that, say, protons also have a probability distribution for where they might be found — but if you take that to be the "shape of the proton", then you completely lose the ability to talk about the proton's internal structure, with the three quarks floating around and exchanging gluons. The proton does have a shape, sort of, that being the shape given by the positions of the quarks, and definitely not given by the probability density function for position of the proton as a whole.
- So the correct answer to "what is the shape of the electron?" is "no one knows; it might not have one at all", but is definitely not "the shape of its probability density function". --Trovatore (talk) 09:48, 9 January 2013 (UTC)
- Regarding the proton shape: You've still got the same problem as with an electron, insofar as quarks are point particles like electrons, which themselves obey quantum rules and thus have the same problems with concepts like localizability and volume. So, since the "shape" of a proton is dependent on the "shape" and "position" of quarks (which is essentially as meaningless a concept as it is with electrons), so I don't think you can meaningfully discuss the shape of a proton any more than of an electron. Even the nucleus of the atom has a structure and a shape that defies easy definition. The smallest objects whereby shape takes on specific, definable, meaning are atoms and molecules, and even there there is some "fuzziness" (i.e. various ways to define atomic radius). --Jayron32 13:39, 9 January 2013 (UTC)
- Well now, hold on. The position of a quark is not "meaningless", it's just quantumly weird. The proton is a quantum superposition of infinitely many states in which all three quarks have precise positions, and each of those states has a precise shape (a triangle, though not the same triangle for each of the states), and where each of the quarks is, as far as anyone knows, a point mass. Anyway I agree that it's the same issue; that was kind of my point. I was explaining why the shape of the orbital is not correctly identified with the shape of the electron. --Trovatore (talk) 18:50, 9 January 2013 (UTC)
- Wait a moment there! Take any three points in space and just try to position them so they aren't in a triangle! That's not "a precise shape"! Literally any position they might be in would be a triangle. (Albeit a "degenerate" one if they lay on the same straight line or two or more of them were at the same position).
- I think that was my point. --Trovatore (talk) 20:32, 9 January 2013 (UTC)
- Wait a moment there! Take any three points in space and just try to position them so they aren't in a triangle! That's not "a precise shape"! Literally any position they might be in would be a triangle. (Albeit a "degenerate" one if they lay on the same straight line or two or more of them were at the same position).
- A proton certainly has a size although what it is precisely isn't clear.[1] And there are people looking at the shape of electrons.[2] Sean.hoyland - talk 19:04, 9 January 2013 (UTC)
- Just glanced at it — is there anything there that's inconsistent with the hypothesis that the bare electron is a point mass? If so I didn't see it. --Trovatore (talk) 19:07, 9 January 2013 (UTC)
- No, not yet but I posted that in response to the comment "The smallest objects whereby shape takes on specific, definable, meaning..." to show that people are looking at the shape of smaller objects. Sean.hoyland - talk 06:27, 10 January 2013 (UTC)
- I'm not sure what those people measured but it has nothing to do with the pointlike nature of electrons. Saying that the electron is pointlike means that it's coupled to other fields individually at each point of spacetime (in the Standard Model Lagrangian). In contrast an extended object could interact with other objects at different spatial locations at the same time. Any evidence of that kind of nonlocality would be huge news, much bigger than the Higgs discovery. (It's an expected feature of quantum gravity, though, at least in string theory, since strings aren't points. But no one expects to be able to detect their size.) -- BenRG (talk) 03:32, 12 January 2013 (UTC)
- No, not yet but I posted that in response to the comment "The smallest objects whereby shape takes on specific, definable, meaning..." to show that people are looking at the shape of smaller objects. Sean.hoyland - talk 06:27, 10 January 2013 (UTC)
- Just glanced at it — is there anything there that's inconsistent with the hypothesis that the bare electron is a point mass? If so I didn't see it. --Trovatore (talk) 19:07, 9 January 2013 (UTC)
- Well now, hold on. The position of a quark is not "meaningless", it's just quantumly weird. The proton is a quantum superposition of infinitely many states in which all three quarks have precise positions, and each of those states has a precise shape (a triangle, though not the same triangle for each of the states), and where each of the quarks is, as far as anyone knows, a point mass. Anyway I agree that it's the same issue; that was kind of my point. I was explaining why the shape of the orbital is not correctly identified with the shape of the electron. --Trovatore (talk) 18:50, 9 January 2013 (UTC)
- The strong force field of protons and neutrons is well approximated by a sphere with a diameter of about 1 fm within which the three "valence" quarks move freely. Electrons are point particles in a sense, but don't forget that the Standard Model is a quantized classical field theory. The quantization leads to pointlike behavior but there's a sense in which electrons can spread out even at the classical level. But unlike nucleons they have no intrinsic size as far as anyone can tell. -- BenRG (talk) 17:32, 9 January 2013 (UTC)
- Regarding the proton shape: You've still got the same problem as with an electron, insofar as quarks are point particles like electrons, which themselves obey quantum rules and thus have the same problems with concepts like localizability and volume. So, since the "shape" of a proton is dependent on the "shape" and "position" of quarks (which is essentially as meaningless a concept as it is with electrons), so I don't think you can meaningfully discuss the shape of a proton any more than of an electron. Even the nucleus of the atom has a structure and a shape that defies easy definition. The smallest objects whereby shape takes on specific, definable, meaning are atoms and molecules, and even there there is some "fuzziness" (i.e. various ways to define atomic radius). --Jayron32 13:39, 9 January 2013 (UTC)
- My point is that it's probability envelope is probably the best way to represent it, as that at least has some geometric definition. See atomic orbitals for some others. StuRat (talk) 09:37, 9 January 2013 (UTC)
- I'm not sure electrons have a "shape", per se. As far as I know, no one has ever demonstrated that they are anything but pure geometrical points (though of uncertain position). Whether the question even makes sense at all might depend on your interpretation of quantum mechanics.
- However, if they do take up space, what shape should they be, except a sphere? Why prefer one direction over another? (They do have spin, so I suppose you could argue for some anisotropy based on the spin vector.) --Trovatore (talk) 09:25, 9 January 2013 (UTC)
The whole discussion revolves around electrons, most of which is beyond my knowledge. The questions about astronomical objects are not completely answered. Please clarify it. Parimal Kumar Singh (talk) 04:58, 10 January 2013 (UTC)
- Solids are generally held together by chemical bonds. This is the force that gives structure to a rock or an asteroid for example. For small to medium sized solid bodies, the force of gravity is too small to substantially change this. However, once a solid body gets very large (i.e. > 1000 km or so), the gravitational force starts to overcome the chemical bonds and pull it into an approximate sphere. On planets, mountains are held together with the same basic forces that hold together a rock. In general, the larger the planet gets, the greater the gravity at its surface, and the smaller the size the mountains that can exist without gravity breaking the rock apart and causing the structure to collapse. For stars and gas giants, there isn't a solid surface at all. Gravity simply pulls on the gas and it flows towards the center until the pressure of underlying gas can exert an equal outward force. Because the gravitational force is essentially uniform in all directions, the gas also ends up uniformly distributed (i.e. spherical), with small corrections if the object is rotating. Dragons flight (talk) 11:58, 10 January 2013 (UTC)
- Essentially, one can appeal to symmetry. Large objects are only held together by gravity. Gravity is a force that acts equally in all directions - so material will end up uniformly distributed - and the only shape that has this perfect symmetry is a sphere. However, if the sphere is rotating (as is the case for almost all heavenly bodies) - the centrifugal force will be stronger at the points furthest from the axis of rotation - resulting in slight bulge around the equator. So none of these bodies are perfectly spherical.
- That said, real planets are incredibly close to being perfect spheres. At smaller scales, such as mountains and such - the atomic forces holding the rock together is stronger than gravity - so more complicated, asymmetrical, shapes become possible. Despite that, the earth is more perfectly smooth and round than a regulation billard ball - and the atmosphere and oceans are thinner than a layer of paint at that same scale. SteveBaker (talk) 14:54, 10 January 2013 (UTC)
Capacitor article, what does Pand Vc stand for?
[edit]In the article Capacitor section "Energy of electric field" what does and stand for in the specified formulas? Electron9 (talk) 09:16, 9 January 2013 (UTC)
- P stands for (electrical) power, i.e. how much energy is used per unit of time (in Watt or J/s); Vc is simply the voltage over the capacitor. - Lindert (talk) 13:14, 9 January 2013 (UTC)
- I updated the article part you had added, Electron9. DMacks (talk) 15:57, 9 January 2013 (UTC)
- How do you know that P is for power? Electron9 (talk) 16:48, 9 January 2013 (UTC)
- Its use as "P=IV" and related equations seems to meet the definition of Electric power. DMacks (talk) 16:53, 9 January 2013 (UTC)
- Where do you see "I" defined as a variable for current in the article? I did think exactly like you until I found it depended on other undefined variables. Electron9 (talk) 19:44, 9 January 2013 (UTC)
- Yes, I was assuming standard symbols there. But also "Power = work per unit time", which seems consistent with the integral form. DMacks (talk) 19:52, 9 January 2013 (UTC)
- Where do you see "I" defined as a variable for current in the article? I did think exactly like you until I found it depended on other undefined variables. Electron9 (talk) 19:44, 9 January 2013 (UTC)
- Its use as "P=IV" and related equations seems to meet the definition of Electric power. DMacks (talk) 16:53, 9 January 2013 (UTC)
Strength of gravity anomalies on earth
[edit]
The image File:GRACE globe 1.png shows the differences in gravitational acceleration on different parts of Earth, after normalizing the differences caused by the rotation of earth and the polar radius being smaller than the equational radius. But the image doesn't include a legend for the colors, so I can't tell what the red and blue colors represent. So my question is, how large are these variations in the gravitational acceleration? – b_jonas 11:23, 9 January 2013 (UTC)
PS. the image description links to [3]. I tried to look there, but a quick look didn't give an answer. – b_jonas 11:25, 9 January 2013 (UTC)
- There is a version of that same image in our the article: Gravity Recovery and Climate Experiment that includes the legend:
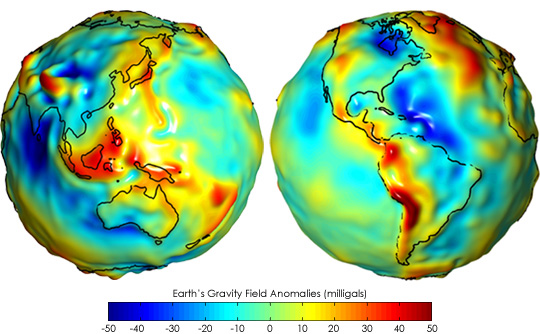
- I have no idea why that was cropped out in the version you refer to!
- The scale is in milligals - and the numbers range from -50 to +50, which is 50 thousandths of a 1 cm sec-2 acceleration...normal gravity is around 9.8 m sec-2 and these variations are at most -/+ 0.0005m sec-2 - which is really very tiny! Note that gravity varies by a half percent between equator and pole - so what your latitude is matters vastly more than whether you're standing on a red spot or a blue spot on that map! SteveBaker (talk) 13:45, 9 January 2013 (UTC)
- Thank you for the answer. – b_jonas 20:01, 9 January 2013 (UTC)
AL Amyloidosis Prognosis
[edit]This site states that there is a 40 month prognosis with AL Amyliodosis, siting a British Medical Journal, without speaking to advanced symptomatic organ failure which accompanies most diagnosis. The actual prognosis of AL Amyliodosis with Cogestive Heart Failure (CHF) is 4-6 months. As it can take 4-6 months for a CHF patient to recieve the Amyliod diagnosis, siting a 40 month prognosis without additional information may lead patients and their families to adopt a wait and see posture early in the discovery process, thus leading to patient mortality. Perhaps someone would like to address this. — Preceding unsigned comment added by 71.232.106.77 (talk) 13:01, 9 January 2013 (UTC)
- You really need to point this out and discuss it on the Talk:AL amyloidosis page where people discuss the content of that article. This is the reference desk - we answer questions and do information searches and such but we don't generally edit articles for people. Right now, I can tell you that the editors of that article are going to want to know where you got your information from. They need a reliable source that they can mention in the article...right now, they're going to say that the British Medical Journal is a highly respected source of medical information - and if it says 40 months - then that's the number they're going to put into the article unless there is another, more recent, reliable source that says otherwise. If you can point to a research paper or some kind of study document - then that will help them to get the article sorted out. But please discuss this on the talk page of AL amyloidosis - the reference desk is not the right place. SteveBaker (talk) 13:24, 9 January 2013 (UTC)
Electronic configuration of copper and chromium
[edit]Electronic configuration of copper, chromium, and some other elements are different from what it should be. Why is it so ? Show your knowledge (talk) 13:49, 9 January 2013 (UTC)
- Because the rubric we learn for predicting the electron configurations is an approximation of reality, and reality is much more complex. --Jayron32 14:08, 9 January 2013 (UTC)
- A bit more: The Aufbau principle (aka Madelung's Rule) is a very rough approximation indeed, as it isn't really based on a rigorous mathematical understanding of the quantum mechanics of how electrons interact with the nucleus and with each other to produce a specific configuration. Instead, it is designed as a very rough "rule of thumb" that will get most people the right answer most of the time. There are other, more accurate, approximations , such as the Hartree–Fock method. So the answer is that the reason why copper, chromium, palladium (and indeed many other elements) don't directly obey the Aufbau principle is that the Aufbau principle is wrong, but it's right enough for first year chemistry students to get most elements correct. Indeed, for anyone that never gets to rigorous computational quantum mechanics, the Aufbua principle + memorize the exceptions is usually as far as they will ever get. --Jayron32 14:22, 9 January 2013 (UTC)
- You used the word aka, what does it mean ? I still don't understand the reason behind my question. Show your knowledge (talk) 15:16, 9 January 2013 (UTC)
- Sorry. Aka is an abbreviation for "also known as". The answer to your question is that the method you were taught for determining electron configurations is wrong. Better methods exist, but they involve the sorts of mathematics that 99% of people (indeed, that the majority of chemists themselves) never learn. All methods are wrong (as the quote goes "All models are wrong, but some are useful"), but the one taught you in your chemistry class is more wrong than others. It's right enough, however, for the purposes of your chemistry class, and teaching you less wrong models would require taking several years to teach the mathematics first. --Jayron32 15:24, 9 January 2013 (UTC)
- You used the word aka, what does it mean ? I still don't understand the reason behind my question. Show your knowledge (talk) 15:16, 9 January 2013 (UTC)
- A bit more: The Aufbau principle (aka Madelung's Rule) is a very rough approximation indeed, as it isn't really based on a rigorous mathematical understanding of the quantum mechanics of how electrons interact with the nucleus and with each other to produce a specific configuration. Instead, it is designed as a very rough "rule of thumb" that will get most people the right answer most of the time. There are other, more accurate, approximations , such as the Hartree–Fock method. So the answer is that the reason why copper, chromium, palladium (and indeed many other elements) don't directly obey the Aufbau principle is that the Aufbau principle is wrong, but it's right enough for first year chemistry students to get most elements correct. Indeed, for anyone that never gets to rigorous computational quantum mechanics, the Aufbua principle + memorize the exceptions is usually as far as they will ever get. --Jayron32 14:22, 9 January 2013 (UTC)
Thank you Jayron, your second explanation was excellent. According to what have been taught in my chemistry class: In chromium second last shell and last shell contain 5 and 1 electrons respectively. On the other hand, second last shell and last shell of copper contain 10 and 1 electrons respectively. Is this true in reality ? Show your knowledge (talk) 15:49, 9 January 2013 (UTC)
- Yes, that is really true. The electron configuration of every element can be determined spectroscopically, such that you can experimentally determine the actual configuration of electrons in an element. The "rules" you are taught in chemistry class (the Aufbau principle) whereby you add electrons to each element based on a simple formula, is mostly right, but it gets certain elements (like Chromium and Copper) wrong, insofar as the Aufbau principle predicts a configuration of 4s23d4 for chromium, but actual experiments have determined that the ground state configuration is actually 4s13d5. There are models better than the Aufbau principle that closer match reality (i.e. models that actually predict the correct configuration of chromium and copper rather than explain them away as "exceptions" to the rule) but those models require a level of mathematics which is well beyond the average first year chemistry student. --Jayron32 16:03, 9 January 2013 (UTC)
I read this in a book: "The deviation (from Aufbau principle) in electron configuration of some elements is because completely filled (d10, f14) or completely half-filled (d5, f7) configurations are more stable. The stability is due to two factors. One, these configurations are more symmetrical which increases their stability. In symmetrical arrangements, the electrons are farthest away from each other, and their mutual shielding is the minimum. The coulombic repulsive forces between them are also weakest. Due to both these reasons, the electrons are attracted more strongly towards nucleus. Two,the electrons in degenerate orbitals can exchange their position. These exchanges also increase the stability. In completely filled and completely half filled orbitals, the number of such possible exchanges are maximum which make such electron configurations more stable." Is this explanation correct ? Show your knowledge (talk) 05:32, 10 January 2013 (UTC)
- Sure. That's pretty much it. There's also probably some small effects from spin-spin coupling of like-spin electrons as well, and larger atoms start to have relativistic effects which affect their configurations. Calculating the exact energy contributions of all of these various effects is quite messy, which is why they don't teach it to you right away, for the most part the Aufbau principle works, except for the "half-filled d" and "all-filled d" exceptions of the copper and chromium groups. There's also other exceptions besides those (there's actually upwards of two dozen of them, at least), and not all of them so easily follow the "half-filled/all-filled" rule-of-thumb, i.e. Ruthenium, Palladium, Cerium, and several more. --Jayron32 06:11, 10 January 2013
(UTC)
You used this sentence There are other, more accurate, approximations , such as the Hartree–Fock method. This means that there are also some other methods to get electron configuration of an element. Can you name some of them ? Show your knowledge (talk) 11:33, 12 January 2013 (UTC)
The exceptions do not matter in real chemistry. These elements have many low-lying excited states that may easily become the ground states in chemical environments (the excitation energies to the Aufbau-predicted "correct" configurations are well within the range of possible chemical bond energies). You will not go terribly wrong even if you mentally have the wrong ground-state configuration in mind for Cr and Cu etc., especially since those ground-state configurations are for gaseous atoms sitting alone by themselves, so don't have much to do with real chemistry. Double sharp (talk) 04:41, 3 June 2020 (UTC)
Why are all milk frothing mugs made out of stainless steel?
[edit]Does milk froth better in a stainless steel container than in a container made of another material (like ceramic)? If so, WHY does milk froth best in stainless steel? — Preceding unsigned comment added by Rmravicz (talk • contribs) 14:16, 9 January 2013 (UTC)
- I assume that you are referring to coffee machines. My home one came with a plastic frothing jug, but I broke it, so now I use a Pyrex one. Both frothed equally as well as the stainless steel ones used in coffee shops. I suspect stainless steel is used, as it is easier to clean than glass or plastic and it won't break when dropped. I also think the professional jugs are doubled walled, so the Barista can froth enough milk for several cups and keep it hot. --TrogWoolley (talk) 15:24, 9 January 2013 (UTC)
Three questions
[edit]1) Why is oxygen necessary for survival? Why not other gaseous element?
2) What make our body to trap only oxygen when we breath in?
3? How long does it take to digest hen meat in our stomach?
Sunny Singh 14:21, 9 January 2013 (UTC) — Preceding unsigned comment added by Sunnysinghthebaba (talk • contribs)
- 1) The reaction of oxygen with carbon based molecules creates a lot of energy, which we need for our organs and muscles to operate. There is no commonly available gas on this planet apart from oxygen that will perform this function.
- 2) Our blood vessels contain proteins (hemoglobin) designed to bond with oxygen, thus 'trapping' it, while leaving the nitrogen etc alone. However, they also bond with carbon dioxide and carbon monoxide, so that's why these gases are dangerous in significant concentrations because they take the place of oxygen and so deprive our organs of oxygen. - Lindert (talk) 14:37, 9 January 2013 (UTC)
- The reaction of oxygen with carbon also creates carbon monoxide. Sunny Singh 06:01, 10 January 2013 (UTC)
- (ec) 1) Because our bodies produce energy by combustion/respiration, which is an oxidation reaction, and thus requires oxygen. More generally, our biology is based on carbohydrates and proteins, both of which contain significant amounts of oxygen. Nitrogen is also needed, but N2 molecules are very hard to break - no regularly occurring substance burns in nitrogen - so we allow nitrogen-fixing plants (and the animals that eat them, unless we are vegans) to get hold of the nitrogen for us. The other atmospheric gases that come to mind are carbon dioxide, which is a product of respiration, and thus cannot be effectively used in such a reaction, and argon, which as a noble gas is almost (but not quite) impossible to react with anything.
- 2) The structure of haemoglobin in the blood allows for oxygen atoms to be attached; this takes place in the lungs (roughly speaking). It's not true that we trap only oxygen: for example, carbon monoxide poisoning takes place because the CO molecules bond more readily to the haemoglobin than the oxygen does, and won't come off.
- 3) As I recall, food (of whatever sort) doesn't stay in the stomach itself more than a couple of hours or so, but takes 24-36 hours to traverse the entire gastrointestinal tract. Digestion takes place throughout most of this period, by several methods. PS: in English, the meat of the hen is referred to as 'chicken'. AlexTiefling (talk) 14:43, 9 January 2013 (UTC)
- (edit conflict) 1) Oxygen is involved in numerous biological processes, Dioxygen in biological reactions covers some of them. But basically, oxygen works by reacting with other molecules to release energy. You can see this dramatically in most combustion reactions, where the oxygen combines with things violently and rapidly to release huge amounts of heat very quickly. In biology, similar reactions are mediated by many enzymes which allow for a slow controlled release of energy that allows every other biological process to occur. 2) Oxygen is trapped from the air by hemoglobin, the specifics of which are covered in the Dioxygen in biological reactions article, in the "Oxygen uptake and transport" section. 3) Digestion#Human digestion process covers the specific timing of various parts of the human digestive process, including the amount of time food spends in the stomach. --Jayron32 14:46, 9 January 2013 (UTC)
I got the answers of Q. 1 and 2. In Q.3 I was asking for time in hours. Sunny Singh 11:52, 12 January 2013 (UTC)
nanotube compressed air storage
[edit]So, nanotubes supposedly have such high tensile strength just a few molecules wide it can support a space elevator.
Anyhoo forget nanotubes. I'm not sure why I mentioned it. Imagine a sheet of zero weight and infinite tensile strength, and you make a container out of this. You are allowed to pump in air at a small differential to the atmosphere and some kind of mechanism gets it inside, and similarly you are allowed to get air out at the rate you want, magically. However, the container does not have further magical properties, and it is subject to normal laws of thermodynamics. We are just not concentrating on the tensile properties and the mechanism of letting you pump into it and out of it.
So, in this case, what is the limiting factor of how much air you could store in this for use as a mobile power source? Is it the weight of the air? Is it how hot the air inside would get? What happens if, for example, you put a million or a billion cubic feet of air inside?
Basically, I am trying to imagine what happens at the limit of a "perfect container" for storing compressed air.
Now let's turn to practicalities. Practically, what happens to the pressure as you put more and more air in? Where do current known materials have a theoretical limit on how much pressure they can hold? How does the weight of material needed increase in response to the amount of pressure you want? Does something special happen when so much air is put in that the pressure melts it? Freezes it? (What I mean, is would the pressure suddenly spike immensely when you put more air in once the air inside is already liquid, or already solid?)
Forgive me, I assume higher pressure compresses by first liquidizing then freezing the air. Please correct me if I'm wrong.
Now let's turn to the pump. What are the practical limitations on trying to create a pump that you can "easily" pump into and "practically" get power out of in non-explosive bursts, but rather a rate that is more appropriate for driving a car?
Basically, I am trying to imagine what would happen if a nuclear powerplant tried to load a container with several inches of nanotube sheeting with enough air to power a car for ten years. What /would/ happen?
I know I've asked a lot of questions - thanks for any responses!!! --91.120.48.242 (talk) 14:47, 9 January 2013 (UTC)
- In the real world, the energy that can be stored in a compressed gas is limited by the material strength of the container. If you assume infinite container strength and a really, really good pump, eventually the container will contain enough mass to hold together without a container. This might be a neutron star or a black hole or it could be a gas giant -- it depends on the size of the container. Also see Bottled gas and Pressure vessel. --Guy Macon (talk) 16:13, 9 January 2013 (UTC)
- Well before that, the material would be too heavy to transport. Are you saying that with the above assumptions ("infinite container strength and a really, really good pump") the package is already a lot better than gasoline? (Since we can simply stop once we've pumped enough energy into it for it to surpass the energy densitity of gasoline?) 178.48.114.143 (talk) 16:36, 9 January 2013 (UTC)
- You might be interested in the articles Compressed-air vehicle and Compressed air energy storage. In practice, without cooling the container during compression, more and more of the pump energy will go to heat the compressed air (and hence the container, until it melts, or until all the energy of the pump is going into heating the atmosphere round the container). Using cooling, the compressed air can be converted into a pale blue liquid, and that's about as far as it is practical to go since most liquids are very difficult to compress further. Dbfirs 17:33, 9 January 2013 (UTC)
- Existing compressed-air cars use air stored at about 50Mpa of pressure and achieve about 1/20th of the energy density of gasoline (so 1/20th of the range for similarly sized "fuel" tanks). You might want to check out Orders of magnitude (pressure) to see what the effect of 1000Mpa pressure would be to get the same range as a gas-powered car. It's considerably higher than (for example) the pressures used to cut steel using high pressure water jets! Actually using that much pressure would be difficult because releasing a 1GPa air flow would destroy anything it hit! To get the pressure up enough to where it could store enough energy to run a car for a year, you're up at pressures where your carbon nanotubes would spontaneously form diamonds and your oxygen would turn into a metal!
- Seriously...this isn't going to happen! SteveBaker (talk) 18:09, 9 January 2013 (UTC)
- It's funny you say that, because I decided to look you up. In the Orders of Magnitude (Pressure) article you linked, 50 Mpa is listed next to 10^7 (the exact exponent would be 7.6), and 1000 Mpa is thus 10^9 (the exact exponent). Well guess what is listed under 10^9: "tensile strength of Inconel 625 according to Aircraft metal strength tables and the Mil-Hdbk-5". So, your very own link shows that storing air at 1000 Mpa is just fine, as long as it's stored in Inconel. So tell me: why not load an Inconel container with 1000 Mpa of air? As for the pump, you can simply make it pneumatic, and out of Inconel, like this: a long tube that tapers. You can then wrap it around the pump or whatever. I've uploaded a diagram. http://i.imgur.com/ksZVB.jpg . I'm not sure if it will work, but I'm sure you'll tell me. I don't see why not... — Preceding unsigned comment added by 91.120.48.242 (talk) 09:28, 10 January 2013 (UTC)
- Inconel 718 is tough stuff - and it is actually used for cryogenic storage tanks - so it's properties in the realms you're interested in are well explored. The stuff is incredibly hard to work with in manufacturing - so the cost of such things would be incredibly high...but our article notes that it can be cut with a Water jet cutter - which you'll note cannot yet reach 1000 Mpa - topping out at maybe 600 to 700 Mpa. So Inconel isn't a magic pressure-resisting-material that you can just assume can be used to make the various parts of your contraption - it's just not that simple!
- Moreover, the best pumps available for water jet cutters can only reach 700 Mpa - and that limit is going to apply to your car tank filler. Even if a 1000 Mpa pump, storage tank and turbine were available - the cost of building something like that out of Inconel would be extreme! That's a really expensive, exotic material...and it's very hard to machine - so even if it were possible, the cost of producing a pneumatic car with similar range to a $10,000 Kia would be spectacular - and because this is a fundamental limitation of the material - it's not likely to reduce with large-scale production or technological advances. So your idea - even if possible - is never going to be practically useful.
- But the other problem (which earlier respondants have mentioned - but you've failed to consider) is that air liquifies at around 200 atmospheres - which is 20Mpa - so once you get beyond 20 Mpa, you're trying to compress an incompressible liquid - getting even 10% more liquid into that tank would require pressures vastly larger than 1000 Mpa. Hence you can't be using air if you want a car with a longer range than a couple of miles...which is about what real pneumatically powered cars get.
- Then you go on to suggest that you can store enough air to run the car for a year - and that's vastly beyond even this possibility.
- Steve, thank you for this thoughtful response. In fact the "limitation" (once air is liquid, pumping more in becomes exponentially more difficult I gather - could you show me this on a chart?) was what I first asked about. So, really, your statement was misleading where you suggested "You might want to check out Orders of magnitude (pressure) to see what the effect of 1000Mpa pressure would be to get the same range as a gas-powered car." Because you're saying that, in fact, 1000Mpa are not enough to get the same range as a gas-powered car, because shortly after the 50 Mpa we currently use, the contents are all liquid and increasing the pressure from 50 Mpa to 1000 Mpa hardly gets you a greater volume of gas stored. (If I understand what you are now saying correctly). One thing you did not address however is my pneumatic pump idea: you say "Moreover, the best pumps available for water jet cutters can only reach 700 Mpa - and that limit is going to apply to your car tank filler." But I showed you a diagram that is suppossed to act as a funnel. Does it not work this way? If not, why not? The key insight here is that you don't care about the internal pressure of the 1000 Mpa or 800 Mpa or whatever, you just need it output over a large surface. So although you can store it at such an internal pressure, you can get it out over a large surface, gradually. What would happen if, for example, my diagram had a sturdy pump balloon over it (like a pump) with a one-way valve letting it draw air in from the the outside, you squeezed it over a large area, then you let it draw air again. It would look like this: http://i.imgur.com/StAED.png - single arrows are pressure, double arrows are movement. At the left of the three, the pump is neutral. To get to the middle, squeeze the pump: it now expels over a LARGE surface area into the snail. Once you've finished squeezing it, you release. The one-way valve gets snapped shut and you get to the third of the three pictures in which air streams back into it as it expands due to its rubbery shape. (Or it can be pulled open again mechanically.) What do you think?
- For hydrogen this works and is near production. See [4]. However, it depends on specific properties of the nanotube interacting with hydrogen to stabilize it - it's not just a tiny pressure tank, it actually sucks up the hydrogen, more or less. Wnt (talk) 19:46, 9 January 2013 (UTC)
- Oh, yeah - FYI: [5] says that the diameter of the carbon nanotube cable needed to support a space elevator would need to vary between one and 16 centimeters...not "a few molecules wide" as you assert. Indeed one of the most cited scholarly papers on the subject ([6]) concludes with "It is the author’s opinion that the cable, if realized as designed today, will break.". It's strongly likely that carbon nanotubes are incapable of supporting such a structure at any diameter. SteveBaker (talk) 14:44, 10 January 2013 (UTC)
- Right, which is why in part this wasn't really a question about carbon nanotubes as a magic weight-free substance that obeys physics but can store any pressure. I was interested in aspects such as the heat, and the fact that the result would still have to weigh something, and also we have just found out that pumping into it at arbitrary pressure might not be that easy or fun. 91.120.48.242 (talk) 16:05, 10 January 2013 (UTC)
- I still like the thought experiment where you assume perfectly strong materials and an infinitely powerful pump. After all, the original question did specify a container of "infinite tensile strength." Let's assume that you compress your [whatever - it doesn't matter what you start with] down to a tiny, tiny sphere of pure neutronium. Then you let it expand a bit at a time to power your vehicle. And you use that handy infinite-strength material to stop it from falling through the bottom of your car and heading for the earth's core.
- Neutronium has some advantages as a fuel. Because it contains no protons, it is not radioactive. Because it has no electrons, it is totally chemically inert. I am not even going to guess at the hardness or tensile strength. And when it expands you can, in theory, have it expand into any element. I want my car to create gold bars as the exhaust.
- The main disadvantage is that Neutronium is that it is incredibly dense: 4*10^17 kg/m^3 (four times ten to seventeenth power kilograms per cubic meter) -- about a million million times heavier than lead. So you need your chunk of neutronium to be very tiny, otherwise your vehicle will weigh many tons. And I am not sure whether I want to be on the same planet if it ever expands all at once. --Guy Macon (talk) 21:31, 10 January 2013 (UTC)
- I guess people are either taking my "hypothetical" version too far or not far enough. Not far enough when they get bent out of shape about how many PSI we can pump or the PSI that our current materials support: I asked you to assume we could pump, and assume that a weightless material supports any psi. However, they take it too far when they then think that we can get to a neutron star, since I expressly said that my experiment should have NO other special properties: it doesn't insulate, it doesn't make your material weightless, it doesn't prevent the exhaust, etc etc. So, I would think that at the point where I place my mental experiment, where hte ONLY three things we elide are: 1) the pump mechanics; assume we can pump at any PSI into the container; 2) the container itself's weight, 3) the container itself's ability to ohold pressure, which are assumed to be infinite, 0, and infinite respectively. This assumption still leaves you having to deal with the weight of the gas, as well as with any phase transitions as pressure comes out of the pump, and with the temperature problems. Under these circumstances, how much "weight" could we ACTUALLY (asssuming 3 magic assumptions) pump into a car? Surely no more than a few tons. So, how much energy does a few tons (say, ten tons) of air represent when compressed into a gas tank mobile size. What PSI does that get us. How much do we have to deal with the thermal problem? 91.120.48.242 (talk) 07:42, 11 January 2013 (UTC)
- Your assumptions allow us to pump gas in until the tank is full of neutronium. Sorry if you don't like where your assumptions lead. Now you are asking us to pretend that your assumptions don't allow us to pump gas in until the tank is full of neutronium, and to pick some other limit. Sorry, but the answer is still that your assumptions allow us to pump gas in until the tank is full of neutronium. (Actually, they allow the formation of a black hole, but you said earlier that you wanted to extract energy by allowing the material in the tank to expand, so i am ruling out a black hole.) --Guy Macon (talk) 19:17, 12 January 2013 (UTC)
- Okay, this is fine. Could you show me how it becomes more difficult to continue pumping inside as we go from gas inside to liquid and solid or metal or whatever? Is it just the case that with each milliwatt-hour (or whatever unit) of compressive energy that you pump into, the pressure spikes by a lot more than it did before the phase change? I would like to see this diagram for, for example, air. THanks. 178.48.114.143 (talk) 23:25, 12 January 2013 (UTC)
What is the difference between anode rays and canal rays ?
[edit]Last paragraph of article Anode ray says As these perforations were named as canal so these rays are called canal rays. These are not the anode rays as these were not originated from the anode. The article doesn't clearly describes how these two rays are different. Please help ! Want to be Einstein (talk) 15:03, 9 January 2013 (UTC)
- This terminology (and this technology) is not in common use in this century. So, it's really a matter of historical interest to quibble over the finer points of this terminology. Our article already links to a reprint of a 1913 article, Rays of positive electricity, published by J. J. Thomson; you can rest assured that he used his terminology consistently. As with all scientific jargon, different scientists have used different variations; but as of today (2013), not many people at all are actively publishing descriptions of these types of devices using that terminology. Most books and papers today will talk about charged ion beams or electron beams, and will talk about the positive and negative particles, or device terminals, (rather than the more confusing "anode" and "cathode" terminology). You'll still hear the term "Cathode Ray Tube" in common use, because those devices had such an important historical impact; but you won't be hearing about CRTs for very much longer, as almost every single use for vacuum-tubes - from display monitors to klystron microwave amplifiers to Bremsstrahlung x-ray sources - have all been replaced by better semiconductor or solid-state alternatives. Nimur (talk) 19:34, 9 January 2013 (UTC)
- Every single use, Nimur? Not for a good while yet. To generate microwaves for cooking, at continuous power levels of several hundred watts, nothing beats a cavity magnetron, a type of cathode ray tube that is nothing more than a heated cathode surrounded by a shaped anode. Want to know what is the cheapest, most stable, easiest way to accurately (say 3- to 4-digit accuracy) measure voltages 30 kV and up? A triode valve (preferably a tungsten filament valve) - a type of cathode ray tube! You use the anode as the input, earth the grid, and vary the cathode voltage (typically 10 to 20 V or so) to get a specific cathode current (microamps or less), virtually none of which flows in the grid. The relationship of anode voltage to cathode voltage at a specific current is precisely linear within a good working range, dependent only on the tube physical geometry. The relationship to current is well understood (the "three halves power rule"). At lower voltages, or high voltages at lower accuracy, voltmeters normally use multiplier resistors. However, getting accurate stable resistors for 20 kV and up is not trivial - usually 2 digit accuracy is as good as you can get. There's also the old gold leaf electroscope, but they have woefull accuracy and are not linear. There's other niche applications for vacuum tubes/cathode ray tubes too. Keit 121.215.151.39 (talk) 23:44, 9 January 2013 (UTC)
- Valid points. High-power solid-state devices are encroaching, but have not totally replaced tubes in some applications. By 2025, I suspect you'll even see microwave ovens using switch-mode IGBTs instead of cavity resonators. And I am not alone in this speculation: Power Electronics magazine published a technical article on 100 kW solid-state (IGBT) converters - so there's hardly any power- or frequency- regime that is "off-limits" for semiconductors. Pick up your favorite microwave or RF newsletter, and you'll see higher- and higher-power RF implemented in silicon. You are correct; the highest power systems haven't made the switch, yet. But it's a matter of time. Solid-state power is cheaper, safer, easier, better, more efficient, ... all the reasons that all other industry applications have switched to semiconductors. Nimur (talk) 00:09, 10 January 2013 (UTC)
- Every single use, Nimur? Not for a good while yet. To generate microwaves for cooking, at continuous power levels of several hundred watts, nothing beats a cavity magnetron, a type of cathode ray tube that is nothing more than a heated cathode surrounded by a shaped anode. Want to know what is the cheapest, most stable, easiest way to accurately (say 3- to 4-digit accuracy) measure voltages 30 kV and up? A triode valve (preferably a tungsten filament valve) - a type of cathode ray tube! You use the anode as the input, earth the grid, and vary the cathode voltage (typically 10 to 20 V or so) to get a specific cathode current (microamps or less), virtually none of which flows in the grid. The relationship of anode voltage to cathode voltage at a specific current is precisely linear within a good working range, dependent only on the tube physical geometry. The relationship to current is well understood (the "three halves power rule"). At lower voltages, or high voltages at lower accuracy, voltmeters normally use multiplier resistors. However, getting accurate stable resistors for 20 kV and up is not trivial - usually 2 digit accuracy is as good as you can get. There's also the old gold leaf electroscope, but they have woefull accuracy and are not linear. There's other niche applications for vacuum tubes/cathode ray tubes too. Keit 121.215.151.39 (talk) 23:44, 9 January 2013 (UTC)
- You are certainly correct in saying vacuum tube technology is obsolete for most applications - that is very obvious. However niche applications for tubes continue. IGBT's are inherently low frequency devices and will not replace magnetrons. If you read the article about switch mode power conversion you linked to, you'll see they think 50 kHz is a good achievement. The triode valve method of measuring extra high voltages works well for short (microsecond) pulses too - nothing else does. Keit 124.178.60.57 (talk) 00:21, 10 January 2013 (UTC)
(Edit Conflict)
- (Note: every technical explanation on the Internet must by law contain at least one glaring error that makes the writer look like an idiot...)
- Start with a bunch of atoms in the form of a gas.
- Cosmic rays and natural radioactivity knock electrons off, making (a few) ions - atoms that are missing an electron.
- Apply a high voltage across the anode and cathode (typically made of metal)
- High voltage accelerates ions toward the negative cathode.
- Ions hit atoms in gas.
- This knocks electrons off of atoms, making many more ions - a chain reaction
- High voltage accelerates ions toward the negative cathode.
- So we have ions traveling from the anode to the cathode - Anode rays.
- Meanwhile the electrons travel from the cathode to the anode - Cathode rays.
- (You can also get electrons from a heated cathode, but that's another story.)
- In 1886 when Eugen Goldstein was first figuring this out, it was hard to look at the anode rays and cathode rays going past each other both ways and figure out which was which.
- Goldstein put small perforations (canals) in the cathode.
- Some of the ions in the anode rays passed though the canals.
- Now he had a ray that was just the ions from the anode ray, with no electrons from the cathode ray to confuse the issue.
- He called these rays that left the cathode in the opposite direction as the cathode rays (but only if the cathode had "canals") "Canal Rays" and the name stuck.
- And yes, our article explains this poorly. Canal rays only appear to originate in the "canals". Then again, few engineers talk about "rays" or "cathodes" anymore. We talks about Electrons, Ions, negative terminals and positive terminals. Still, you see the old terms in a lot of the early papers from the scientists who discovered this stuff. --Guy Macon (talk) 19:53, 9 January 2013 (UTC)
- "(Note: every technical explanation on the Internet must by law contain at least one glaring error that makes the writer look like an idiot...)" - the entire Reference desk system depends on this fact to keep us all interested! SteveBaker (talk) 20:16, 9 January 2013 (UTC)
Buoyant force
[edit]Suppose an object with flat base placed in a container containing water and container also has flat base. The object has rectangular shape and its density is lower than water. The object is submerged (by hand) into water in such a way that its base touches the base of the container. In that situation, there is almost no water below the object and also there is whole water of the container above object and the weight of this water column lies on the object. When the hand submerging the object is removed from water, the object comes on the surface of water as it has lower density than water. The question is:Why does object come on the surface even when there is no water below object to push it up and also the object is forced down by the weight of water column above it ? Does buoyant force depend on the amount of water present below an object ? I tried my best to explain where I got stuck, thank you for answering my question. Sunny Singh (DAV) (talk) 16:48, 9 January 2013 (UTC)
- The pressure of the water is proportional to depth, and any air trapped below the object will exert the appropriate pressure under the object for that depth (being greater than the pressure on the top of the object at a lower depth). If you succeed in producing a seal round the edges of the base, and allow a tiny amount of flexibility so that the trapped air can expand, then the pressure under the object may well become lower than that on the top, so the object will not rise but "stick" to the bottom of the container. I think the same could happen to a very small quantity of water if inrush of water below the object is prevented. Dbfirs 17:09, 9 January 2013 (UTC)
- There is always going to be some water under the flat-bottomed object - you can't get a perfect contact between those two objects. But just do the thought-experiment of removing the water and just pushing the two objects together in air. If there was a perfect seal between the two objects, they'd be held together by the air pressure just like a suction cup on a sheet of glass! The deal in water is just the same. But if there is *any* gap, then water will intrude and because pressure in a liquid or gas exerts a force equally in all directions - there is nothing to prevent the object from floating away. SteveBaker (talk) 17:54, 9 January 2013 (UTC)
- Aquaplaning or hydroplaning might be relevant. A very very tiny amount of water - just a few drops! - can be put into circumstances such that it has a very high dynamic pressure. In those cases, those few drops of water are sustaining the weight of an entire object above them - sometimes resulting in pressures equivalent to many thousands of PSI! That's enough to lift the entire weight of a heavy object - like a truck or a car or a four-hundred-ton cargo aircraft. This is a problem worthy of an entire NASA facility, the Aircraft Landing Dynamics Facility at NASA Langley. Here are some fun videos of this research: NASA Research on Hydroplaning. All this trouble comes from the fact that water is almost totally incompressible. Just a few drops of water will still occupy the same volume even if they are put under the crushing weight of thousands of atmospheres of pressure. Almost no water, if suitably constrained by a container, is therefore able to buoyantly lift a very very heavy object. Nimur (talk) 19:08, 9 January 2013 (UTC)
- The same is, of course, true of the bottom of the very very heavy object. Ultimately there is only one layer of atoms at the bottom of anything, yet they hold it up. I think that SteveBaker's thought experiment works very well. Consider an object "stuck" to the bottom of a pool because there is a suction cup or other "vacuum" under it. What makes that a vacuum is that it is lower pressure relative to the water at the very bottom of the pool; in other words, we normally expect that pressure to propagate through perfectly. I suppose what amazes most is that physics is so good at "doing the math" - in other words, that if you have a narrow tube filled with atoms of water constantly moving about, that somehow the water at the near end and at the far end manage to come into such perfect agreement about what the pressure really is. The same amazement strikes people with other events, such as the ability to do DNA hybridization of a primer with a specific sequence out of an entire genome - as we see in that case, the computational power, though extraordinary, does have limits. Still, it is pretty amazing when you consider that the molecules doing the "math" by smashing together are moving only somewhere on the order of the speed of sound (I'm not entirely sure about the relation of this to the thermal speed according to the Boltzmann constant). Wnt (talk) 19:43, 9 January 2013 (UTC)
- The point is that although they are only moving at the speed of sound, they are moving an incredibly short distance until they hit something and transfer the information. So the number of collisions per second is an ungodly high number - and that's how come this "calculation" happens so fast. SteveBaker (talk) 20:13, 9 January 2013 (UTC)
- A related concept is Choked flow in a hydraulic or pneumatic system, where opening up a valve further downsteam has no effect on flow upsteam because there is a section of pipe with supersonic flow between them. It's the pneumatic/hydraulic equivelant of the speed of light; no information can travel faster than the speed of sound in the gas/liquid, so the upstream system doesn't "know" you opened the valve downstream. --Guy Macon (talk) 20:16, 9 January 2013 (UTC)
- The point is that although they are only moving at the speed of sound, they are moving an incredibly short distance until they hit something and transfer the information. So the number of collisions per second is an ungodly high number - and that's how come this "calculation" happens so fast. SteveBaker (talk) 20:13, 9 January 2013 (UTC)
- The same is, of course, true of the bottom of the very very heavy object. Ultimately there is only one layer of atoms at the bottom of anything, yet they hold it up. I think that SteveBaker's thought experiment works very well. Consider an object "stuck" to the bottom of a pool because there is a suction cup or other "vacuum" under it. What makes that a vacuum is that it is lower pressure relative to the water at the very bottom of the pool; in other words, we normally expect that pressure to propagate through perfectly. I suppose what amazes most is that physics is so good at "doing the math" - in other words, that if you have a narrow tube filled with atoms of water constantly moving about, that somehow the water at the near end and at the far end manage to come into such perfect agreement about what the pressure really is. The same amazement strikes people with other events, such as the ability to do DNA hybridization of a primer with a specific sequence out of an entire genome - as we see in that case, the computational power, though extraordinary, does have limits. Still, it is pretty amazing when you consider that the molecules doing the "math" by smashing together are moving only somewhere on the order of the speed of sound (I'm not entirely sure about the relation of this to the thermal speed according to the Boltzmann constant). Wnt (talk) 19:43, 9 January 2013 (UTC)
- Depth won't matter, in fact the deeper it is, the less stable it is.. If the object maintains it's volume and the density is less than water, it will float. Extreme depth submarines used gasoline (incompressible fluid that is less dense than water). There is no "floating force", rather gravity pulls on the denser water harder than on the object. The system is in unstable equilibrium with the less dense object on the bottom and will revert to a lower energy state of stable equilibrium. Think of a rod pinned in the middle with a heavy ball at one end and a lighter ball at the the other. Trying to make the lighter ball stay at the bottom is difficult because the slightest perturbation will cause it to swing to a lower energy state. The same is true for your block in a water column. It is statically in equilibrium, but any perturbation will cause the system to revert to the lowest energy state which is the denser water beneath the floating block. It is directly analogous to mechanical equilibrium. There is a static solution where the block can remain at the bottom, it's just not a stable solution and the system will revert to the lowest energy state --DHeyward (talk) 09:29, 10 January 2013 (UTC)
- I thought that Sunny Singh was asking about forces. The "static equilibrium" of your claim will occur only when water is prevented from flowing underneath. This is different from your example of unstable equilibrium. I see why you made the comparison, but I don't think it helps to answer the question (unless I misunderstood the question, of course). Dbfirs 21:02, 10 January 2013 (UTC)
Butterfly flight
[edit]this is totally from left field, based on a single observation: a cabbagemoth butterfly was flying right to left - a stiff breeze arose, blowing in the opposite direction - but the butterfly continued unperturbed, whereas one would expect it to be blown away.
Possibly the air current wasn't wide enough to reach both me and the butterfly - but possibly it was. I thought back, and couldn't think of a time when I'd seen a butterfly blown off course. Has anyone here? Have they some as-yet-un-understood mechanism for beating the wind?
Ta
Adambrowne666 (talk) 23:03, 9 January 2013 (UTC)
- I think they just stay grounded during heavy winds. As you said, the wind current you observed must not have reached the butterfly. StuRat (talk) 23:09, 9 January 2013 (UTC)
- Thanks, StuRat, you're most likely right - but let's wait and see if others have made the same observation and we can end up naming a whole new principle after me. Adambrowne666 (talk) 02:37, 10 January 2013 (UTC)
- I live in a suburb in the hills near the edge of a state forest and we get lots of butterflies, I saw easily more then a dozen just the other day while working outside. Nothing about their flight made me think they were not being affected by the breeze, but I always thought butterflies weren't very gracefull fliers even when it's calm. I must admit i wasn't specifically looking for that. I'll make note and watch some butterfiles more closely next time. Vespine (talk) 02:54, 10 January 2013 (UTC)
- Thanks, StuRat, you're most likely right - but let's wait and see if others have made the same observation and we can end up naming a whole new principle after me. Adambrowne666 (talk) 02:37, 10 January 2013 (UTC)
- Thanks, Vespine; maybe they appear to be poor fliers, but are in fact very good. And surely they can't always know when there's about to be a stiff breeze and ground themselves. Let us know if you notice the same thing I did. Adambrowne666 (talk) 12:03, 10 January 2013 (UTC)