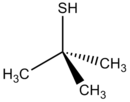
tert-Butylthiol, also known as tert-butyl mercaptan (TBM), and abbreciated t-BuSH, is an organosulfur compound with the formula (CH3)3CSH. This thiol has a strong odor. It is considered a flavoring agent.[1]
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methylpropane-2-thiol | |
| Other names
t-BuSH
2-Methylpropane-2-thiol 2-Methyl-2-propanethiol tert-Butyl mercaptan | |
| Identifiers | |
3D model (JSmol)
|
|
| Abbreviations | TBM |
| ChemSpider | |
| ECHA InfoCard | 100.000.810 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H10S | |
| Molar mass | 90.18 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 0.8 g/mL |
| Melting point | −0.50 °C (31.10 °F; 272.65 K) |
| Boiling point | 62 to 65 °C (144 to 149 °F; 335 to 338 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Preparation
editIt was first prepared in 1890 by Leonard Dobbin[2] by the reaction of zinc sulfide and t-butyl chloride.
The compound was later prepared by the reaction of the Grignard reagent, t-BuMgCl, with sulfur to give the corresponding thiolate, followed by hydrolysis.[3] This preparation is shown below:
- t-BuMgCl + S → t-BuSMgCl
- t-BuSMgCl + H2O → t-BuSH + Mg(OH)Cl
It is made industrially by the reaction of isobutylene with hydrogen sulfide over a clay (silica alumina) catalyst.[4]
Reactions
edittert-Butylthiol is deprotonated by lithium hydride in an aprotic solvent such as hexamethylphosphoramide (HMPA). The resulting lithium thiolate salt has been used as demethylating reagent. For example, treatment with 7-methylguanosine gives guanosine. Other N-methylated nucleosides in tRNA are not demethylated by this reagent.[5]
tert-Butylthiol reacts with thallium(I) ethoxide to give the thallium thiolate:[6]
- (CH3)3CSH + TlOC2H5 → (CH3)3CSTl + HOC2H5
This thallium thiolate can be used to convert acyl chlorides to the thioester:
- (CH3)3CSTl + RCOCl → RCOSC(CH3)3 + TlCl
tert-BuSLi reacts with MoCl4 with to give the tetrathiolate complex:[7]
- MoCl4 + 4 t-BuSLi → Mo(t-BuS)4 + 4 LiCl
Commercial use and occurrence
edittert-Butylthiol is the main ingredient in many gas odorant blends.[citation needed] It is always utilized as a blend of other compounds, typically dimethyl sulfide, methyl ethyl sulfide, tetrahydrothiophene or other mercaptans such as isopropyl mercaptan, sec-butyl mercaptan and/or n-butyl mercaptan, due to its rather high melting point of −0.5 °C (31.1 °F). These blends are used only with natural gas and not propane, as the boiling points of these blends and propane are quite different. Because propane is delivered as a liquid and vaporizes to gas when it is delivered to the appliance, the vapor liquid equilibrium would substantially reduce the amount of odorant blend in the vapor.
Food and flavor
edittert-Butylthiol had been listed on the European Food Safety Authority (FL-no: 12.174) as a flavor additive. There is no indication of what flavor(s) it may have been used in. It has been removed from this list.[8]
tert-butylthiol as a very minor component of cooked potatoes.[9]
Safety
editThe Threshold limit value (TLV) is 0.5 ppm. tert-butylthiol has an odor threshold of <0.33 ppb.[10]
See also
edit- Butanethiol (butyl mercaptan)
- Ethanethiol (ethyl mercaptan)
References
edit- ^ "tert-butyl mercaptan". thegoodscentscompany.com.
- ^ Dobbin, Leonard (1890). "On tertiary Butyl Mercaptan". Journal of the Chemical Society, Transactions. 57: 639–643. doi:10.1039/ct8905700639.
- ^ Rheinboldt, Heinrich; Mott, Friedrich; Motzkus, Erwin; A. D. McMaster; B. M. Mattson; S. T. Michel (1932). "Tertiäres Butylmercaptan". Journal für Praktische Chemie. 134 (9–12): 257–281. doi:10.1002/prac.19321340901.
- ^ Schulze, W.A.; Lyon, J.P. & Short, G.H. (1948). "Synthesis of Tertiary Alkyl Mercaptans". Industrial and Engineering Chemistry. 40 (12). American Chemical Society: 2308–2313. doi:10.1021/ie50468a019.
- ^ Ho, Tse-Lok; Fieser, Mary; Fieser, Louis (2006). "Lithium 2-methylpropane-2-thiolate". Fieser and Fieser's Reagents for Organic Synthesis. doi:10.1002/9780471264194.fos06530. ISBN 0471264199.
- ^ Spessard, Gary O.; Chan, Wan Kit; Masamune, S. (1990). "Preparation of thiol esters: s-tert-butyl cyclohexanecarbothioate and s-tert-butyl 3α,7α,12α-trihydroxy-5β-cholane-24-thioate". Organic Syntheses. 7: 87. doi:10.1002/0471264180.os061.28. ISBN 0471264229.
- ^ Otsuka, Sei; Kamata, Masato; Hirotsu, Ken; Higuchi, Taiichi (1981). "A Novel Molybdenum Thiolato Compound, Tetrakis(tert-butylthiolato)molybdenum(IV). Preparation and Crystal and Molecular Structure". Journal of the American Chemical Society. 103 (11): 3011–3014. doi:10.1021/ja00401a017.
- ^ "Scientific Opinion on Flavouring Group Evaluation 8, Revision 3 (FGE.08Rev3): Aliphatic and alicyclic mono-, di-, tri-, and polysulphides with or without additional oxygenated functional groups from chemical groups 20 and 30". EFSA. 11 May 2011. Retrieved 15 April 2013.
- ^ Gumbmann, M. R.; Burr, H. K. (1964). "Food Flavors and Odors, Volatile Sulfur Compounds in Potatoes". Journal of Agricultural and Food Chemistry. 12 (5): 404–408. Bibcode:1964JAFC...12..404G. doi:10.1021/jf60135a004.
- ^ Devos, M; Patte, F.; Rouault, J.; Lafort, P.; Van Gemert, L. J. (1990). Standardized Human Olfactory Thresholds. Oxford: IRL Press at Oxford University Press. p. 118. ISBN 0199631468.